TL;DR
- The pharmaceutical artwork approval process is the workflow used to review packaging, labeling, and promotional materials before release.
- Because artwork must meet strict regulatory standards, even small errors can create compliance risks or trigger product recalls.
- Structured approval workflows, checklists, and standardized templates help teams reduce manual errors and speed up the entire process.
- Centralized artwork management platforms allow teams to collaborate on compliant artwork across global markets.
- Modern artwork management tools provide detailed audit trails, version control, and automated checks to help teams ensure compliance.
- Platforms like Filestage streamline artwork approvals by centralizing feedback, tracking versions, and organizing stakeholders across the entire process.
Introduction
When you think of artistic creativity, the pharmaceutical industry probably isn’t the first thing that pops up. But you’d be surprised at how much creative energy goes into designing, developing, and reviewing pharmaceutical artwork.
In the highly regulated pharmaceutical sector, even small artwork mistakes can create compliance risks or trigger costly product recalls.
Because pharmaceutical packaging must meet strict regulatory standards and health authority requirements, every piece of artwork must be carefully reviewed before it reaches global markets.
That’s exactly what I explore in this article.
In simple terms, the pharmaceutical artwork approval process is the workflow teams use to review and approve packaging, labeling, and promotional materials before they reach the market.
Five best practices for the pharmaceutical artwork approval process
The following best practices can help pharmaceutical teams improve their artwork approval workflows, reduce manual errors, and ensure compliant artwork across global markets.
1. Define the key stakeholders
The artwork approval process is a team effort in any industry. But especially pharma. The right group of experts makes sure your artwork meets regulatory, quality, and creative standards.
So, the first step to building a better workflow is to identify your key stakeholders. Here are some teams that should be involved in the process:
- Marketing – in charge of the initial design concept and messaging
- Regulatory affairs – makes sure the artwork meets government standards
- Packaging development – turns the concept into artwork
- Quality assurance – performs quality checks to look for accuracy and artwork design flaws
- External printing partners – produce the final printed packaging materials once the artwork is approved
In many pharmaceutical companies, the artwork approval process is governed by standard operating procedures that define how different stakeholders collaborate throughout the entire process.
Once you’ve identified your stakeholders, it’s time to decide when to involve them in the approval process. Ideally, you should segment your reviewers to make sure the right people approve your artwork at the right stage of the process.
Using pharmaceutical review software like Filestage can help you do this.
Less time chasing feedback, more time on creative strategy
Online proofing software can cut review cycles by 30%, freeing up time for high-stakes work. Read our guide to see if it’s the right solution for your team.
Filestage is designed specifically to streamline and secure the creative review process for companies in regulated industries.
Here’s how your pharmaceutical promotional reviews could look in Filestage:
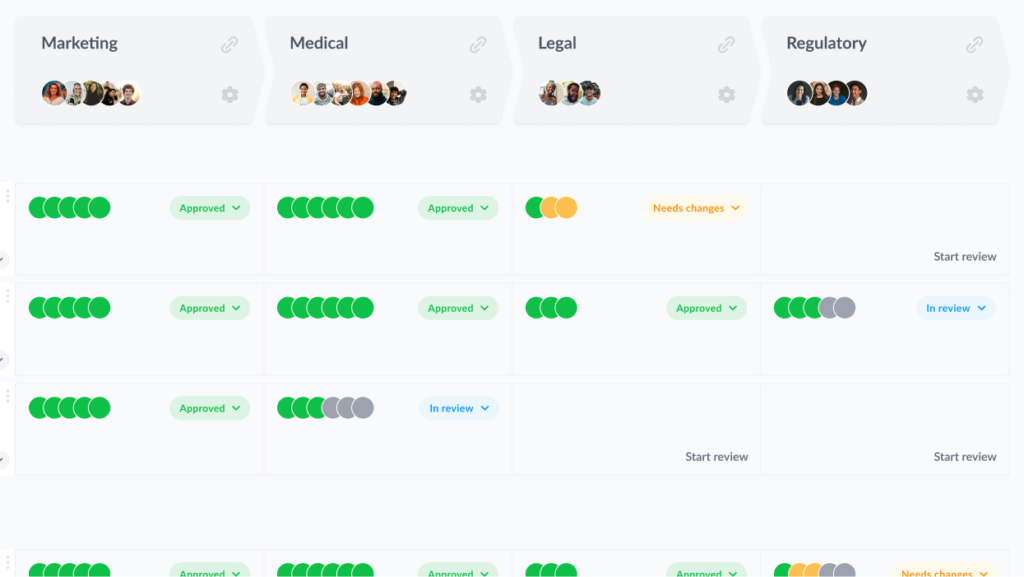
2. Standardize the artwork approval process
Data from the US Food and Drug Administration (USFDA) revealed that in a six-month period, it recorded 455 recall notices. Interestingly, 51% of those were due to mislabeling, and 13% were as a result of faulty packaging.
A standardized artwork approval process provides a reliable framework for reviewing packaging and promotional materials. It will also save you a lot of time and compliance headaches. Trust me.
Using standardized templates, structured approval workflows, and review checklists helps teams reduce manual errors and confirm that compliant artwork reaches the final approval stage.
Here are the main benefits of designing a well-defined workflow:
- Improves consistency and cohesion in your artwork
- Reduces errors and maintains product quality
- Makes sure key review stages are never missed
- Helps you meet regulatory requirements
- Streamlines collaboration with cross-functional teams
- Helps you make sure you’re sending error-free pharmaceutical labels to print
Start by defining a standard process with clear steps and timelines.
A typical pharma artwork approval process includes the following steps:
- Artwork creation – Marketing or design teams develop the initial packaging or promotional artwork based on product information and regulatory guidelines.
- Internal review – Internal stakeholders such as marketing, regulatory affairs, and packaging teams review the draft to check compliance and messaging accuracy.
- Regulatory validation – Regulatory specialists verify that the artwork meets local and international compliance requirements.
- Quality assurance checks – QA teams confirm that labeling, dosage information, and formatting are correct.
- Final approval and release – Once feedback is implemented, the approved artwork is released for printing or distribution.
This structured workflow helps pharma teams move artwork from initial design to regulatory approval and final production without missing critical compliance checks.
After carrying out these steps, you’re ready to build pre-defined templates for artwork submissions. This will make sure you have all the key information from the start of every project.
You can automate this process with Filestage. Once you create a custom workflow, you can save it as a template and reuse it across multiple projects – from packaging to in-store advertising displays.
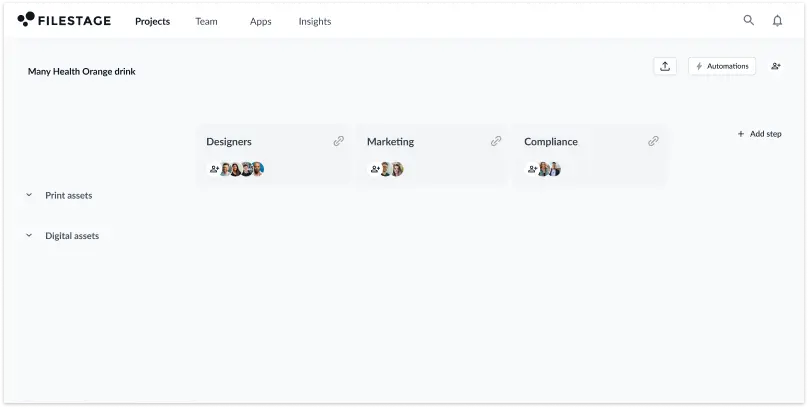
You can also automate time-consuming manual tasks like setting deadlines and moving approved versions to the next review stage, shaving up to 30% off your pharma content approval timeline.

“Filestage saves our company a huge amount of time. Instead of writing detailed instructions over email, reviewers can just click on a file to add comments in context. This makes everything faster – from giving feedback, to managing the process, to making edits before sharing the next version.”
Karina Berner, Creative Production Specialist at Sartorius
3. Understand the specific regulatory requirements
Pharma marketing compliance can be complex to understand, especially if you operate across multiple regions with different standards.
This regulatory complexity is one of the main reasons pharmaceutical artwork approvals require such careful review.
Integrating regulatory experts at the start gives you a better chance of spotting potential compliance issues early on. And it will make sure your brand isn’t responsible for the next big false advertising fail.
Once you understand the regulatory requirements, create checklists and reference materials to guide teams at every stage of artwork creation and review.
Using structured checklists during the artwork approval process helps verify critical elements before sign-off, reducing the risk of errors and regulatory issues.
It may require an initial time investment, but it can significantly cut delays and reworks in the late stages of your project.
4. Manage artwork from a centralized hub
Scattered feedback and versioning chaos are very real symptoms of an ineffective artwork approval process, making it difficult to keep teams and stakeholders on the same page.
Without a centralized artwork system, teams may accidentally work from outdated files, creating confusion and duplicated effort.
Version control problems can result in outdated designs being used in production, increasing the risk of regulatory errors.
Fortunately, there’s a way to reduce these bottlenecks.
Managing the full artwork process in a review and approval platform like Filestage allows you to centralize all assets, feedback, and content versions.
Here’s how you can streamline artwork management with key features from Filestage:
- Create a central repository for content assets and feedback
- Leave in-context feedback directly on packaging artwork (no more neverending email threads)
- Manage old content versions so everyone’s always working on the right iteration
- View versions and comments side by side to confirm feedback has been implemented
- Integrate Filestage with other tools in your tech stack for a seamless experience
Version control is particularly important in pharma, where multiple revisions of packaging artwork may exist across regulatory submissions, internal reviews, and printing partners.
Advanced proofing tools can also detect inconsistencies early in the artwork approval process, helping teams catch errors before they lead to costly reprints or regulatory issues.

Manage every approval in one place
Request approval without sending a single email with Filestage.
5. Measure your performance
Your artwork approval process should evolve as your organization and regulatory requirements change.
As compliance requirements continue to change quickly, you need to check in on your workflows regularly to make sure they’re still optimal. You can do this by looking for bottlenecks and identifying where you need to make improvements.
The first step is to get feedback from reviewers and stakeholders to find out where they’re having issues.
Next, look at the data. Tracking performance metrics across the artwork approval process helps teams identify bottlenecks and improve speed over time.
With Filestage Insights, you can quickly visualize how your artwork review process is performing and gauge your team’s productivity.
Here’s how the dashboard looks:
Why the artwork approval process in pharma matters
The artwork approval process plays a critical role in pharmaceutical packaging and labeling workflows. Pharmaceutical artwork isn’t just about aesthetics. It carries essential product information that must be accurate and clearly presented, with details about usage, dosage, and potential side effects.
Both labeling and pharma packaging artwork play a pivotal role in safeguarding users, helping them make informed decisions about the product.
For example, barcodes and QR codes printed on pharmaceutical packaging must remain scannable and conform to GS1 standards to ensure traceability and product safety.
That’s why medical artwork has to meet strict regulatory requirements that ensure all the information provided is accurate and clear. Regulatory bodies must make sure that every piece of artwork (from the package insert to the label) meets industry guidelines.
With so many reviewers involved, the artwork approval process is complex and time-intensive.
Streamline artwork management with Filestage
Thanks to stringent regulatory compliance standards, quality control is a very real necessity in pharma. An effective workflow streamlines lengthy review processes, keeps all stakeholders aligned, and makes sure your packaging artwork complies with legal requirements. So you can get faster approvals.
Ready to build a better artwork management system? Get a free 30-day Filestage trial today.
FAQ
What is the artwork approval process in pharma?
The artwork approval process in pharma is the workflow used to review and approve packaging, labeling, and promotional artwork before it reaches the market. It typically involves marketing teams, regulatory specialists, quality assurance, and packaging experts working together to verify that all information is accurate and compliant.
Why is artwork approval important in the pharmaceutical industry?
Pharmaceutical artwork contains critical information such as dosage instructions, safety warnings, and regulatory statements. A structured approval process helps prevent labeling errors, reduce compliance risks, and protect patient safety.
What challenges slow down the artwork approval process?
Common challenges include scattered feedback, version control issues, unclear stakeholder responsibilities, and changing regulatory requirements. Without a structured workflow, teams often struggle with delays and repeated review cycles.
How can technology improve pharmaceutical artwork approvals?
Review and approval platforms allow teams to centralize artwork, track versions, collect feedback in context, and manage approvals across multiple stakeholders. Some modern systems also include automated checks or AI-powered tools that verify text accuracy, barcode data, and version consistency before final approval, helping teams detect inconsistencies early in the artwork approval process.